Our Institute
Clinical Trials
Our Science
News
International Patients
- International Patients
- International patient Service Care
- Travel Arrangement and Hospital Admission
- FAQ
- Contact Us
Beneficial treatment of chronic obstructive pulmonary disease (COPD)
based on clonal fetal mesenchymal stem cells (cfMSCs) -
resolution of severe pulmonary fibrosis and substantial improvement in lung function and hypoxia
Organized by Elena, Tingting Wang and Zhengwei Zhang
Chronic obstructive pulmonary disease (COPD) is a progressive pulmonary disorder characterized by impaired expiratory function of the lungs. Clinically, it is often complicated with emphysema and chronic bronchitis, and some patients also present with asthmatic symptoms. Airway inflammation and tissue damage are its core pathological features. Typical symptoms of COPD include fatigue, shortness of breath, and frequent cough with expectoration. The pathogenic root cause lies in excessive bronchial mucus secretion induced by external stimuli, which further leads to bronchial wall thickening and inflammatory responses. Tobacco smoke (active smoking/secondhand smoke) is the primary predisposing factor, and long-term exposure to environmental factors such as dust, air pollution, and chemical substances also significantly increases the risk of the disease. At present, clinical treatment is still focused on symptom control, and there is no radical cure available.
This report shares a first-in-patient clinical case that brings new hope for the treatment of severe COPD: an elderly patient with severe COPD complicated with interstitial lung fibrosis achieved complete resolution of imaging signs of pulmonary fibrosis, accompanied by clinically significant improvement in lung function indicators, and substantial slowdown in disease progression after receiving infusions of a novel stem cell product consisted of purified clonal fetal mesenchymal stem cells (cfMSCs).
I. Patient's Baseline Condition and Disease Progression
A 74-year-old male patient had a 50-year smoking history and was complicated with ischemic heart disease (with a right coronary artery stent implantation). He also had a special genetic trait—high susceptibility to elevated C-reactive protein (CRP) levels, meaning he had an inherently stronger inflammatory response, which also accelerated the progression of his pulmonary disease. The patient's disease progression is as follows (see Figure 1):

Figure 1: Timeline of the patient's disease progression
According to the Global Initiative for Chronic Obstructive Lung Disease (GOLD) classification criteria, the patient was diagnosed with severe COPD (GOLD Grade 3), with obvious airflow limitation, decreased pulmonary diffusing capacity, and a very high risk of respiratory failure.
II. Practical Application Regimen of Stem Cells
In March 2024 and November 2025, the patient received two courses of infusions of clonal fetal thymic mesenchymal stem cell (thymic cfMSCs) products developed by Shenzhen GIMI, as part of a Phase I/II clinical trial (NCT03123458) for the treatment of immune-related diseases in collaboration with participating hospitals. Three intravenous infusions were administered for each course:
First course of infusions: March 4, March 6, and March 8, 2024
Second course of infusions: November 30, December 2, and December 4, 2025
Compared with adult bone marrow/adipose-derived MSCs, the thymic cfMSCs have significant advantages: higher proliferative capacity, lower immunogenicity, stronger paracrine signaling effects, and the ability to exert their functions without long-term in vivo engraftment, making them more suitable for the treatment of inflammatory and fibrotic pulmonary diseases.
III. Efficacy Evaluation
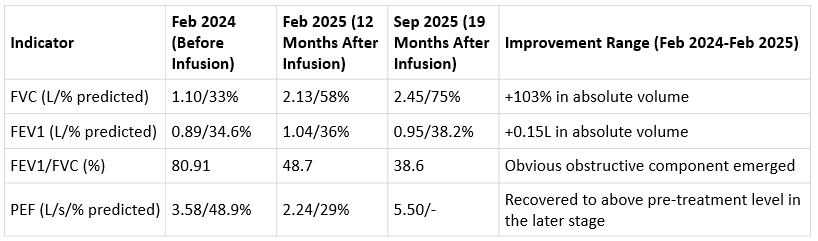
Table 1: Comparison of core lung function indicators before and after the first cfMSC infusions
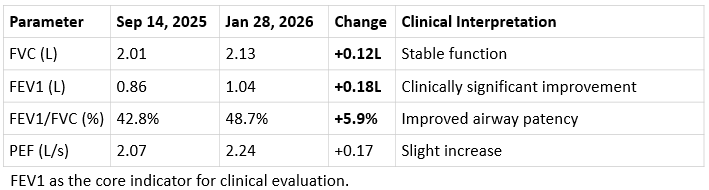
Table 2: Comparison of core lung function indicators before and after the second cfMSC infusions
Definitions of indicators:
*FVC (Forced Vital Capacity): The maximum total volume of gas exhaled forcefully after deep inspiration, reflecting total lung capacity and respiratory muscle strength.
*FEV1 (Forced Expiratory Volume in One Second): The volume of gas exhaled within the first second of the FVC test, directly reflecting airway patency and serving as the core diagnostic indicator for COPD.
*FEV1/FVC (One-second Ratio): The ratio of FEV1 to FVC, used to assess the degree of airflow limitation.
*PEF (Peak Expiratory Flow): The maximum flow rate during exhalation, reflecting the function of large airways.
1. Lung function: substantial increase in core indicators exceeding the clinically significant threshold
Stem cell therapy showed sustained and significant improvement in lung function, with both courses of treatment leading to clear increases in indicators:
After the first course: At 12 months post-treatment, FVC increased to 2.13L and FEV1 to 1.04L, with a 150mL increase in FEV1 compared with pre-treatment; at 19 months post-treatment, FVC further rose to 2.45L, more than doubling the pre-treatment baseline. Despite slight fluctuations in FEV1, it remained consistently above the pre-treatment level, achieving a substantial increase in lung volume.
After the second course: In January 2026 (2 months after infusion, the patient was 78 years old), lung function indicators continued to improve: FVC rose to 2.13L and FEV1 increased significantly to 1.04L, with a 180 mL increase in FEV1 within two months. In clinical COPD research, an FEV1 increase of ≥100 mL is considered clinically significant, and the patient's improvement far exceeded this threshold. Meanwhile, the airway patency indicator FEV1/FVC increased from 42.8% to 48.7%, and PEF rose from 2.07 L/s to 2.24 L/s, indicating a substantial improvement in pulmonary ventilation function.
2.Efficacy comparison: surpassing traditional drugs and pioneering a new path for COPD treatment
To objectively evaluate the efficacy of cfMSC therapy, a comparison was made with Ohtuvayre, the most promising clinical drug for improving FEV1 in COPD at present:
Phase III clinical trials of the drug showed that it increased FEV1 by an average of only about 87–94 mL compared with the placebo, while the patient's FEV1 increase after a single course of stem cell infusion far exceeded this value in this case.
Drug treatment is associated with adverse reactions such as diarrhea, back pain, and hypertension, and its cardiovascular safety requires continuous monitoring. In contrast, stem cell infusion not only achieved better efficacy but also brought about systemic multi-system improvements with no obvious side effects.
3. Imaging: complete resolution of pulmonary fibrosis signs and significant improvement in structural lesions
Chest imaging examinations confirmed that various structural lesions in the patient were significantly relieved and systemic inflammation was effectively controlled:
Pulmonary fibrosis: Nine months after the start of stem cell therapy, a follow-up chest CT showed complete disappearance of previously diagnosed signs of interstitial lung fibrosis, honeycombing changes, and traction bronchiectasis, with no recurrence of the original fibrotic lesions.
Pleural effusion: Thoracostomy tube drainage was required in May 2023; only a trace amount of right basal pleural effusion was observed in December 2024, with no reaccumulation.
Airway abnormalities: Bronchial mucosal thickening, mucus plugs and other manifestations present in May 2023 were completely relieved, and no such findings were mentioned in the imaging examination in December 2024.
Mediastinal lymph nodes: Reduced compared with the previous examination in May 2023 and returned to normal in December 2024.
4. Remarkable efficacy: fibrosis resolution, substantial lung function improvement, and significant slow down in disease progression
After two courses of cfMSC infusion, the patient's disease progression was significantly curbed: before treatment, lung function indicators showed a natural course of severe COPD with continuous deterioration; after stem cell infusion, the rate of FEV1 decline was reduced by approximately 50%, completely reversing the trend of rapid lung function decline and achieving long-term disease stability.
5. Systemic status: reduced dependence on oxygen therapy, improved metabolic homeostasis, and no obvious adverse reactions
In addition to direct improvements in the lungs, the patient's systemic status was comprehensively enhanced: subjective energy levels were significantly improved, sleep quality was better, dependence on oxygen therapy was greatly reduced, and was completely free from the need for a constant oxygen supply. Meanwhile, glucose metabolism indicators tended to be stable, the originally fluctuating hyperglycemia was effectively controlled, and the amplitude of daytime blood glucose fluctuations was significantly reduced. Given that these clinical observations suggested a potential trend of systemic metabolic improvement, an extended metabolomic test was performed 14 months after infusion. The results showed that the patient exhibited an abnormally stable metabolic regulatory state for his age and the severity of his disease.
IV. Significance and prospect of the case: providing a new direction for the treatment of severe COPD
This case is a rare instance of an elderly patient with severe COPD and a genetic predisposition to high inflammation achieving long-term disease stability after stem cell therapy. With the limited efficacy of conventional treatment, cfMSCs achieved comprehensive therapeutic effects including resolution of pulmonary fibrosis, significant improvement in lung function, and slowdown in disease progression through multi-level biological regulation of multiple pathological mechanisms such as inflammation, fibrosis, and metabolism.
Certainly, this case is a single clinical observation, and its results still need to be further verified by larger-sample randomized controlled clinical studies. However, the novel cfMSCs provide a new research direction for such refractory and severe COPD patients, and further confirm the great potential of stem cell therapy in the treatment of chronic pulmonary diseases, laying a solid real-world data foundation for subsequent clinical research and applications.
References
[1] Cona L A. Stem Cell Therapy for COPD in 2023[EB/OL]. 2023-04-28. https://www.dvcstem.com/post/stem-cells-copd.
[2] Broekman W, Khedoe P P S J, Schepers K, et al. Mesenchymal stromal cells: a novel therapy for the treatment of chronic obstructive pulmonary disease?[J]. Thorax, 2018,73(6):565-574.